[ad_1]
middle school biochemist
From an early age, Sikes looked at the world with an insatiable curiosity about how things work. He collected and observed everything from rocks to snakes. “I drove my elementary school teachers crazy,” she says.
In middle school, he was designing experiments to measure chemical reactions in nature, including a toxicology study of the effects of caffeine on sea urchins. He had hoped to persuade his father, who was also a scientist, to soften his coffee habit. While the experiment failed in this regard, it sowed the seed of something greater. Sikes understood how chemistry research could improve health and benefit society.
Although his undergraduate education at Tulane was focused on physical chemistry, Sikes eventually returned to his early biochemical research. At Stanford, where he received his PhD, he began studying redox mechanisms, particularly how certain oxidizing agents attract electrons from other molecules. And he became interested in oxidative stress, which occurs when free radicals in the body (highly reactive molecules that lack one or more electrons that easily oxidize other substances) suppress the antioxidants cells normally produce to neutralize them. This can cause a variety of health problems.
Cancer, in particular, is characterized by higher-than-normal levels of free radicals called reactive oxygen species (ROS). In normal metabolic activity, ROS molecules support cell regeneration and gene expression. However, high ROS production can damage normal cells and facilitate tumor growth.
As a biochemist, Sikes was fascinated by the possibility of detecting and manipulating these changes in cancer cells that doctors are struggling to measure accurately. To see what was happening inside the tumors, he needed to see when the cells were oxidized; turned to fluorescent proteins that emit light of different wavelengths. “We use light-triggered chemistry to detect these redox reactions,” Sikes says.
Turning this into therapeutic potential was only a short step. If doctors can understand the true redox activity underlying a tumor, they can better predict how chemotherapy will stop this activity and allow normal cells to regain control.
Otherwise, they will keep shooting in the dark. Sikes literally had a vision to illuminate his pursuits.
sensors at work
Using their sensors, the researchers could potentially measure when, where, and how much oxidation tumors are experiencing – simply by illuminating them. Fluorescent sensors can also shed light on the mechanisms of action of various therapeutics, helping doctors choose the best for each patient.
Since 2018, Sikes’ team has been collaborating with Tufts pathologist Arthur Tischler to use their biosensors for insight into the redox chemistry behind a variety of cancers. In paper Published in 2020, they investigated the pathology of tumors deficient in succinate dehydrogenase (SDH), an important metabolic enzyme and an inhibitor of ROS production. Low SDH levels have been associated with cancers that are both rare and difficult to treat.
By reconfiguring biochemical processes, it can measure the distinctive chemistry behind almost all aspects of antibody production, tumor development, and human disease.
Using the same biosensors, Sikes and his team were the first to focus on chemotherapies that induce a single oxidizing agent: hydrogen peroxide. In paper In their paper published in Cell Chemical Biology, they outline how they built a sensor specifically designed to detect increased concentrations of hydrogen peroxide, which can selectively kill cancer cells. The team examined 600 molecules as potential therapeutics and identified four that increased hydrogen peroxide in tumor samples.
The team’s success will facilitate clinical trials of new pharmaceuticals. Ideally, the next step is to use these fluorescent sensors to evaluate the effects of these therapeutics in patient-derived tumors.
Fast detection diagnosis
Sikes realized that his technique could also detect pathogens, including the novel coronavirus SARS-CoV-2, which causes covid-19.
To make such a detector, Sikes needed antibody proteins that would react with the virus’s distinctive proteins. But these reactive proteins were absent. That’s why he decided to create them.
In her postdoctoral research, Sikes worked with 2018 Nobel laureate Frances Arnold, a Caltech chemical engineer and a pioneer in creating new proteins with desirable properties.
Sikes’ lab is now designing proteins that lock into prominent folds in proteins characteristic of various pathogens. Engineered proteins emit different wavelengths depending on how they bind with the material of the virus or bacteria.
On the basis of this innovative technology, Sikes has developed rapid diagnostic tests containing a range of reagents that detect one type and exclude others so that healthcare professionals can diagnose infectious diseases more quickly and accurately. His lab focuses on engineering reagents that can identify coronaviruses, respiratory syncytial virus (RSV) and other causes of respiratory disease; Bacteria affecting food safety (especially Listeria and E. coli); and parasitic eukaryotes, for example plasmodiumcauses malaria.
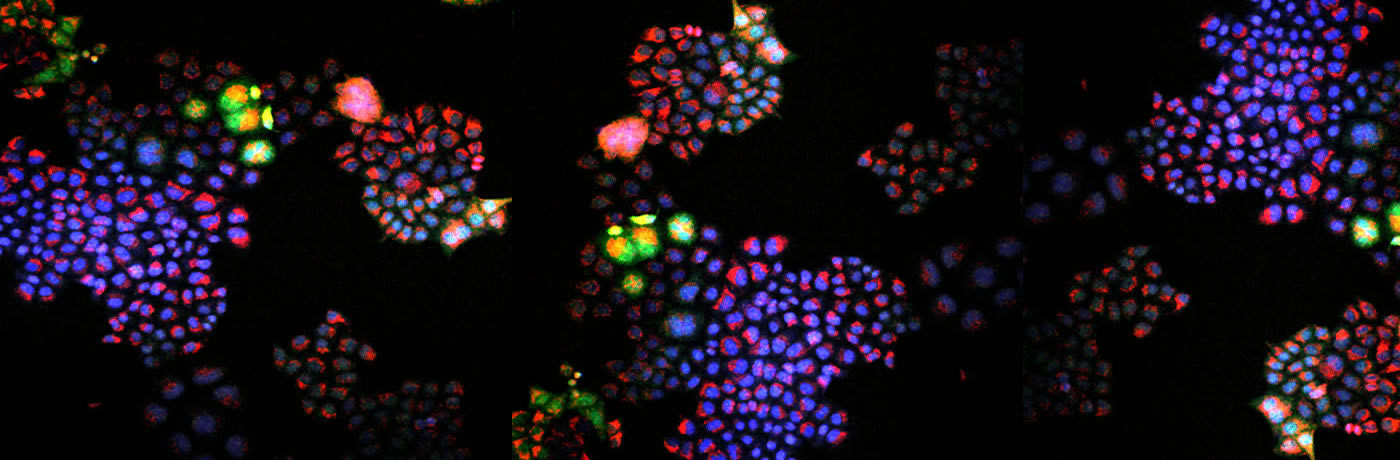
RESEARCHERS’ PERMISSION
Students and postdoctoral researchers at Sikes’ Singapore lab are developing tests that assess immunity to different COVID-19 variants as part of a fast-track research project. As with their other work, the specially designed proteins will react uniquely with each person’s antibody repertoire, allowing the team to better understand the extent and resilience of immunity to COVID at an individual level.
Sikes’ effort to save lives with advancing biosensor technology is only part of its mission to use chemistry research for the benefit of society. He accepted his position at MIT in 2009, largely because of his reputation for research that can be applied to solving social problems. And to further this task, he is seizing opportunities to mentor aspiring scientists.
Each summer, MIT welcomes budding researchers from historically underrepresented fields and schools. Last summer, Sikes mentored students at Spelman College, Morehouse College, and the University of Puerto Rico-Mayagüez. The program provides hands-on opportunities to conduct research and make connections with the Institute’s network of scientists. Sikes also counsels undergraduates at Imperial College London as part of an MIT exchange program.
For Sikes, this is the epitome of what science education should be. “I’m probably learning as much from them as they’ve learned from me,” she says. “I really see this as a collaboration. I’ve been doing this for 20 years… but all these students and postdoctoral students come with their own backgrounds, experiences, and ways of looking at things. Often times they have ideas or hypotheses that I wouldn’t have thought of.”
redox to recover
The mysteries that Sikes has pursued since childhood have all come to a measure: What invisible reactions drive surface phenomena?
Today, by reconfiguring biochemical processes, it can measure the distinctive chemistry behind nearly all aspects of antibody production, tumor development, and human disease. He hopes to finalize biosensor proteins and bring them to market in the next few years, empowering other researchers to improve patient outcomes and mitigate the next pandemic.
This does not mean that Sikes’ lifelong curiosity has been satisfied. There are always other questions to ask. “Hopefully 10 years from now we will be doing something completely different that I can’t even imagine right now,” he says.
[ad_2]
Source link

